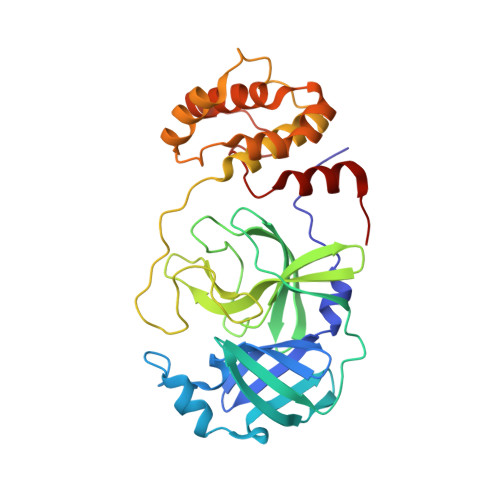
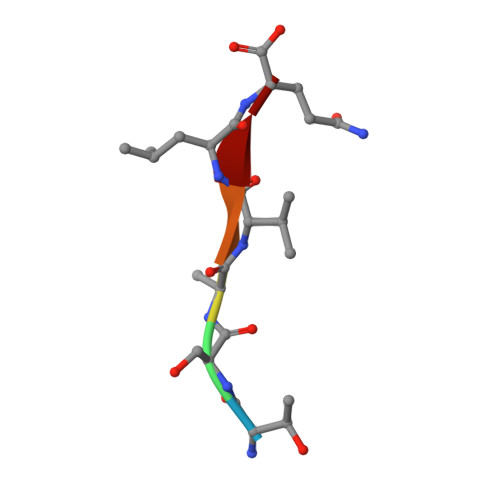
Defining the substrate envelope of SARS-CoV-2 main protease to predict and avoid drug resistance.
Shaqra, A.M., Zvornicanin, S.N., Huang, Q.Y.J., Lockbaum, G.J., Knapp, M., Tandeske, L., Bakan, D.T., Flynn, J., Bolon, D.N.A., Moquin, S., Dovala, D., Kurt Yilmaz, N., Schiffer, C.A.(2022) Nat Commun 13: 3556-3556
- PubMed: 35729165
- DOI: https://doi.org/10.1038/s41467-022-31210-w
- Primary Citation of Related Structures:
7MB4, 7MB5, 7MB6, 7MB7, 7MB8, 7MB9, 7T70, 7T8M, 7T8R, 7T9Y, 7TA4, 7TA7, 7TB2, 7TBT, 7TC4 - PubMed Abstract:
Coronaviruses can evolve and spread rapidly to cause severe disease morbidity and mortality, as exemplified by SARS-CoV-2 variants of the COVID-19 pandemic. Although currently available vaccines remain mostly effective against SARS-CoV-2 variants, additional treatment strategies are needed. Inhibitors that target essential viral enzymes, such as proteases and polymerases, represent key classes of antivirals. However, clinical use of antiviral therapies inevitably leads to emergence of drug resistance. In this study we implemented a strategy to pre-emptively address drug resistance to protease inhibitors targeting the main protease (M pro ) of SARS-CoV-2, an essential enzyme that promotes viral maturation. We solved nine high-resolution cocrystal structures of SARS-CoV-2 M pro bound to substrate peptides and six structures with cleavage products. These structures enabled us to define the substrate envelope of M pro , map the critical recognition elements, and identify evolutionarily vulnerable sites that may be susceptible to resistance mutations that would compromise binding of the newly developed M pro inhibitors. Our results suggest strategies for developing robust inhibitors against SARS-CoV-2 that will retain longer-lasting efficacy against this evolving viral pathogen.
- Department of Biochemistry and Molecular Biotechnology, University of Massachusetts Chan Medical School, Worcester, MA, 01605, US.
Organizational Affiliation: