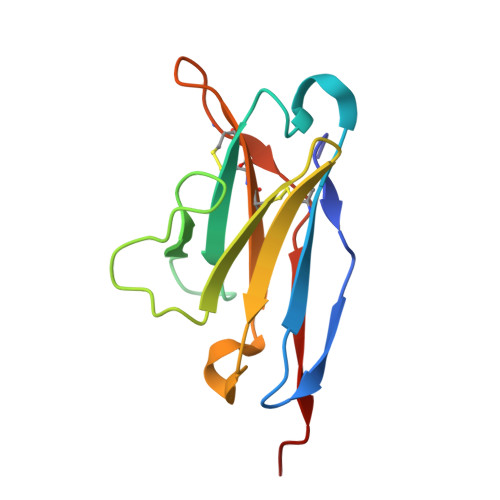
Vaccination of nonhuman primates elicits a broadly neutralizing antibody lineage targeting a quaternary epitope on the HIV-1 Env trimer.
Schleich, F.A., Bale, S., Guenaga, J., Ozorowski, G., Adori, M., Lin, X., Castro Dopico, X., Wilson, R., Chernyshev, M., Cotgreave, A.T., Mandolesi, M., Cluff, J., Doyle, E.D., Sewall, L.M., Lee, W.H., Zhang, S., O'Dell, S., Healy, B.S., Lim, D., Lewis, V.R., Ben-Akiva, E., Irvine, D.J., Doria-Rose, N.A., Corcoran, M., Carnathan, D., Silvestri, G., Wilson, I.A., Ward, A.B., Karlsson Hedestam, G.B., Wyatt, R.T.(2025) Immunity 58: 1598
- PubMed: 40339576
- DOI: https://doi.org/10.1016/j.immuni.2025.04.010
- Primary Citation of Related Structures:
9CU5, 9CU6, 9CV7, 9DMF - PubMed Abstract:
The elicitation of cross-neutralizing antibodies to the HIV-1 envelope glycoprotein (Env) by vaccination remains a major challenge. Here, we immunized previously Env-immunized nonhuman primates with a series of near-native trimers that possessed N-glycan deletions proximal to the conserved CD4 binding site (CD4bs) to focus B cells to this region. Following heterologous boosting with fully glycosylated trimers, we detected tier 2 cross-neutralizing activity in the serum of several animals. Isolation of 185 matched heavy- and light-chain sequences from Env-binding memory B cells from an early responder identified a broadly neutralizing antibody lineage, LJF-0034, which neutralized nearly 70% of an 84-member HIV-1 global panel. High-resolution cryoelectron microscopy (cryo-EM) structures revealed a bifurcated binding mode that bridged the CD4bs to V3 across the gp120:120 interface on two adjacent protomers, evading the proximal N276 glycan impediment to the CD4bs, allowing neutralization breadth. This quaternary epitope defines a potential target for future HIV-1 vaccine development.
- Department of Microbiology, Tumor and Cell Biology, Karolinska Institutet, 171 77 Stockholm, Sweden.
Organizational Affiliation: