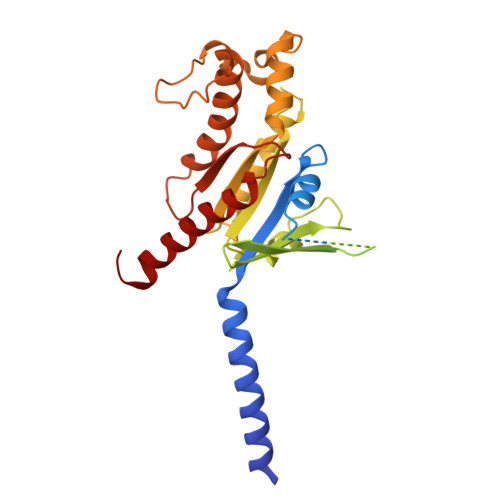
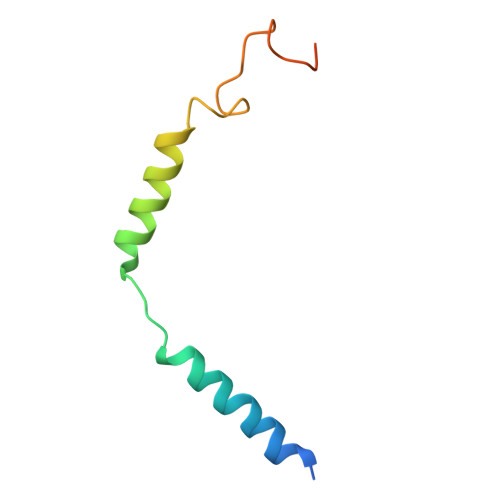
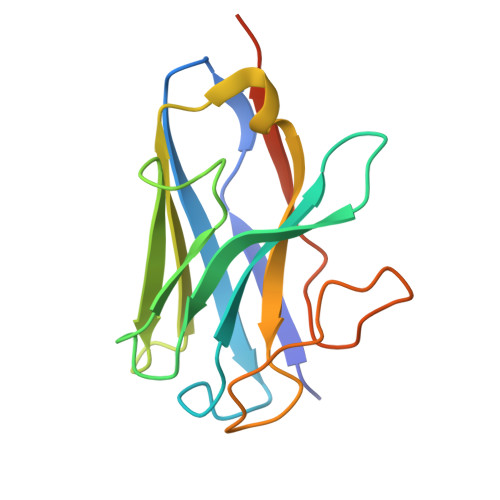
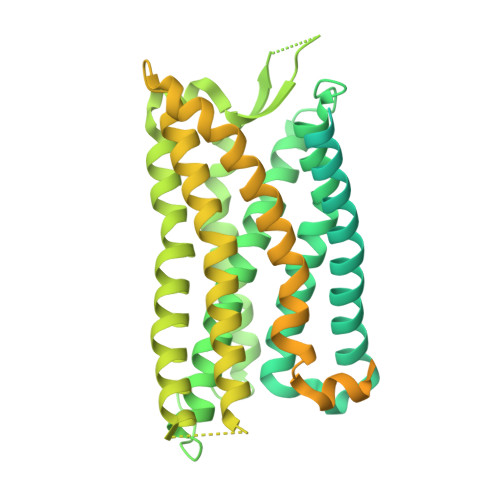
Molecular recognition of two approved drugs Macimorelin and Anamorelin by the growth hormone secretagogue receptor.
Wang, R.L., Sun, J., Liu, H., Guo, S.M., Zhang, Y., Hu, W., Wang, J., Liu, H., Zhuang, Y.W., Jiang, Y., Xie, X., Xu, H.E., Wang, Y.(2025) Acta Pharmacol Sin
- PubMed: 40542284
- DOI: https://doi.org/10.1038/s41401-025-01606-7
- Primary Citation of Related Structures:
9UY3, 9V2N - PubMed Abstract:
The growth hormone secretagogue receptor (GHSR) plays a critical role in regulating growth hormone release and metabolic homeostasis. Understanding the molecular mechanisms of ligand-GHSR recognition is essential for developing therapeutic interventions. In this study, we investigated the molecular recognition mechanisms of two clinically approved drugs: Macimorelin (used for diagnosing adult growth hormone deficiency) and Anamorelin (approved in Japan for cancer cachexia). Using high-resolution cryo-electron microscopy, we determined the structures of GHSR bound to Macimorelin and Anamorelin in complex with G q proteins at resolutions of 2.63 Å and 2.52 Å, respectively. We revealed that both drugs occupied a bifurcated binding pocket divided by a conserved salt bridge between E124 3.33 and R283 6.55 . Through systematic mutagenesis and functional studies, we identified the key residues underlying the higher binding affinity of Anamorelin compared to Macimorelin. In addition, structural comparison of GHSR in complex with different G protein subtypes elucidated the mechanisms driving G protein selectivity. Our results provide crucial insights into GHSR-drug interactions and offer valuable guidance for designing more selective and potent GHSR agonists.
- CAS Key Laboratory of Receptor Research, Center for Structure and Function of Drug Targets, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, China.
Organizational Affiliation: